Contents
Transporting biological samples : what you need to know
Clusters of testing laboratories have significantly increased the frequency and volume of transport of biological and blood samples.
Paragraph 5.4.6. of standard NF EN ISO 15189:2012 on quality requirements and competence applicable to medical biology laboratories states that samples shall be transported “at a temperature specified in the primary specimen collection manual”.
It is necessary to check that this transport is carried out :
- In refrigerated vehicles or insulated transport trunks.
- With a temperature recorder to document this pre-analytical phase.
- Plug and Track designs Plug and Play products, i.e. ready to use.
- We attach great importance to providing you with fast decision-making tools.
- So you can focus on the job you love, and meet your regulatory obligations with complete peace of mind.
Our offer for temperature traceability of sample transports
We offer suitable temperature recorders, in particular the Thermo Button 22L (-40/+85°C, accuracy ±0.5°C, Resolution 0.1°C).
Small and robust, you can slip it closer to the samples in an insulated bag. Many of our customers place the recorder in an empty sample vial, which allows for more accurate reproduction of sample transport conditions.
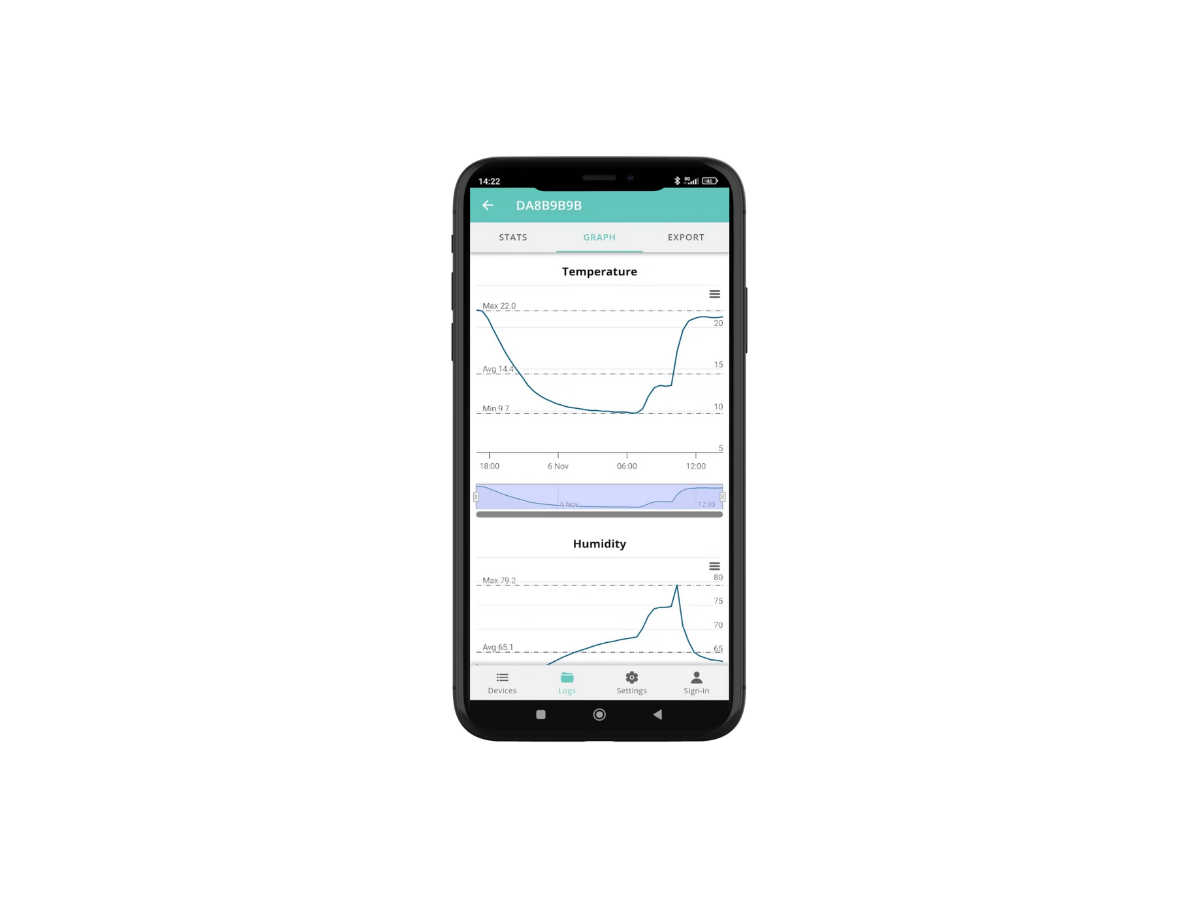
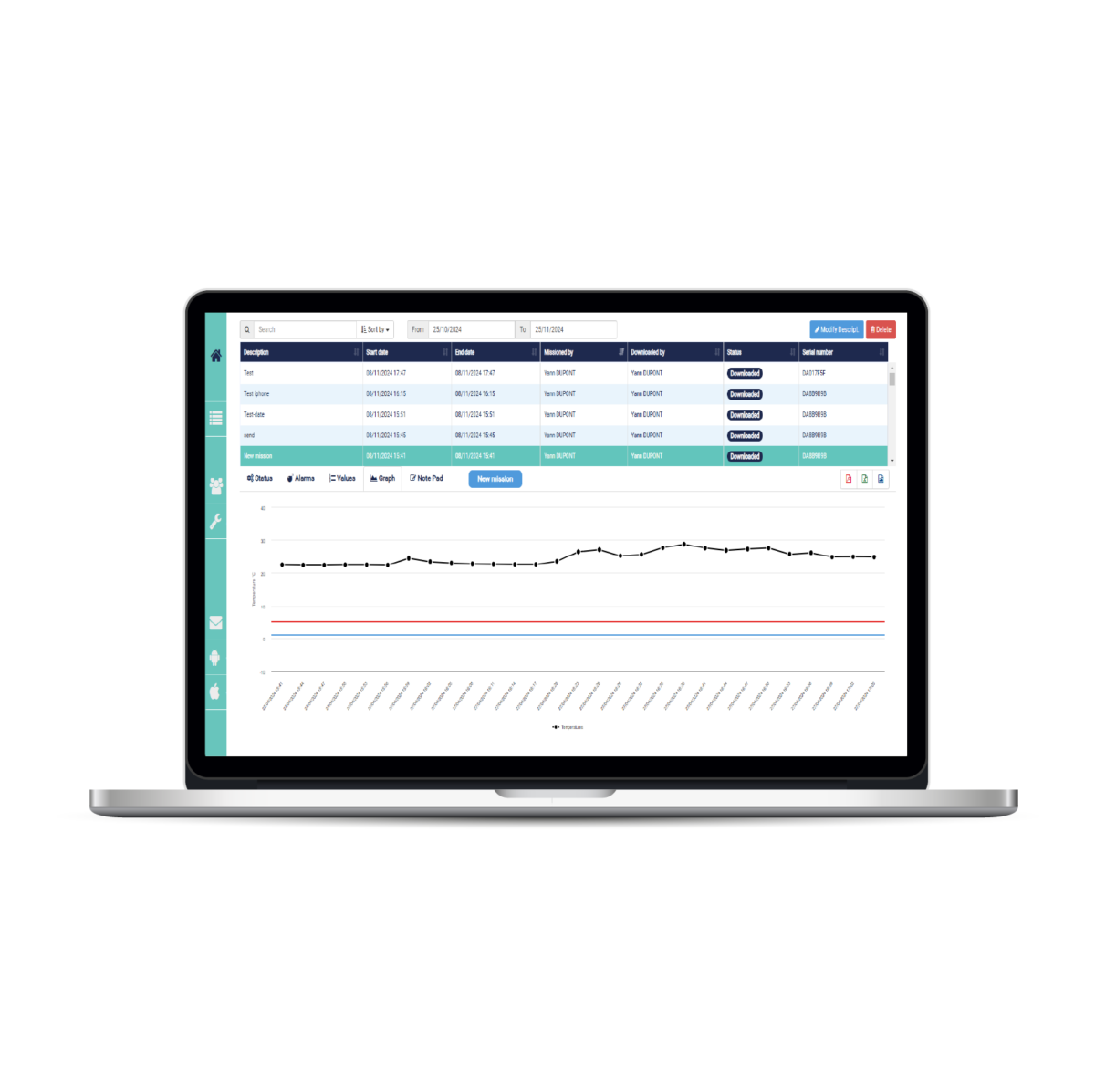
The Thermotrack PC software allows the temperature curve to be printed automatically on receipt of the samples. The archiving and classification of the data is also done automatically by date, vehicle or transport case. You know immediately whether you have respected the cold chain and thus keep a trace for your quality audits.
You can also use Thermotrack Online (web application) to share your data more easily with your different laboratories.
As part of your accreditation, you will need to supplement your equipment with related or ISO 17025 accredited calibration certificates for your Thermo Button dataloggers.